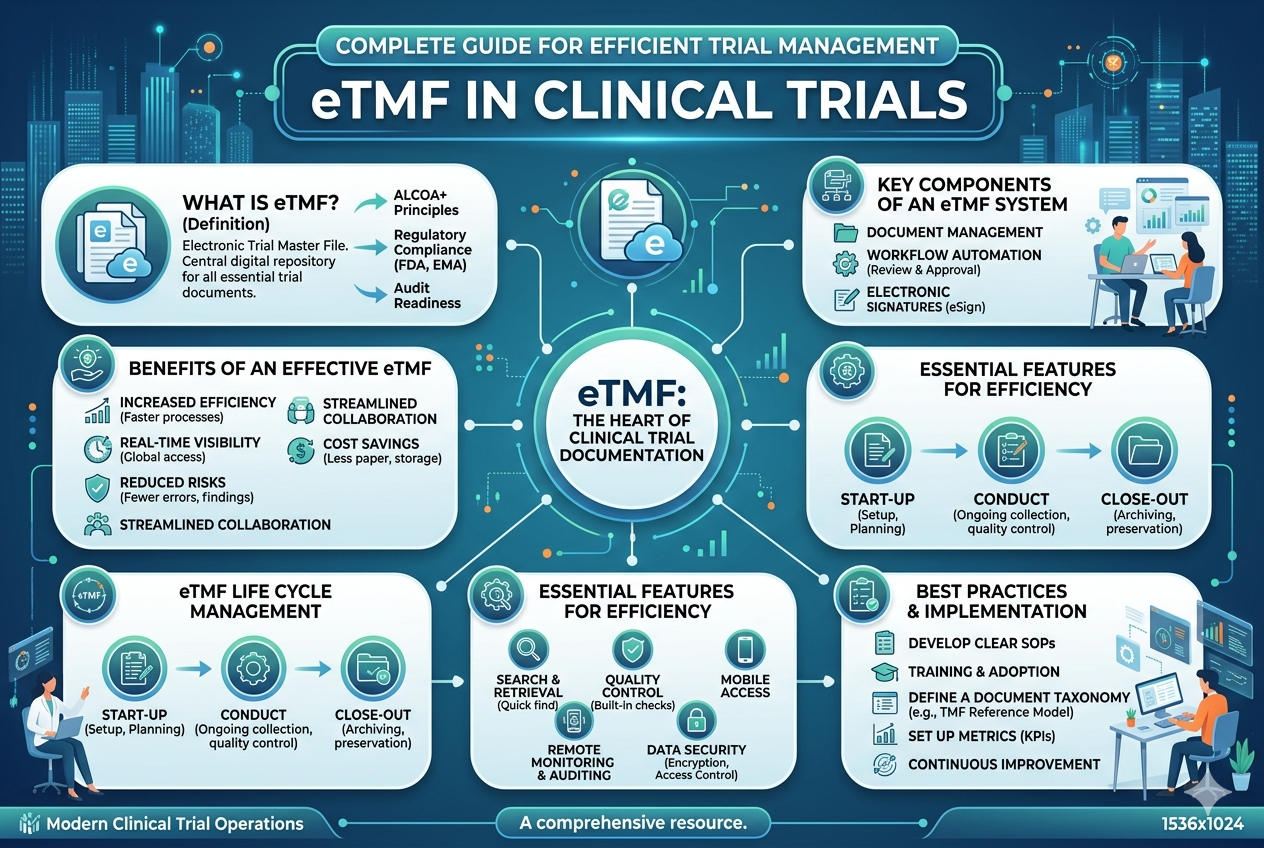
What is an eTMF?
An electronic Trial Master File (eTMF) is a secure electronic trial master file system that collects, organizes, and manages all essential documents related to a clinical trial. It replaces traditional paper-based systems with a centralized digital platform, improving efficiency, accessibility, and compliance.At Yuvainfocare, our ARS eTMF solution is an advanced eTMF software designed to streamline clinical trial document management while ensuring full regulatory compliance and data integrity.
The eTMF acts as documented evidence of how a clinical trial is planned, conducted, and monitored, helping regulatory authorities assess compliance with Good Clinical Practice (GCP).